The CBSE Class 9 Science Syllabus 2025-26 outlines the course content and assessment scheme to be followed in the upcoming academic session. Students must be familiar with the complete syllabus to study the subject in an organized manner and prepare a suitable plan to score well in all tests and examinations scheduled for the current academic session, 2025-26.
General Instructions:
1. There will be an Annual Examination based on the entire syllabus.
2. The Annual Examination will be of 80 marks and 20 marks weightage shall be for Internal Assessment.
3. For Internal Assessment:
a. There will be a Periodic Assessment that would include:
→ For 5 marks- Three periodic tests conducted by the school. Average of the best two tests to be taken that will have a weightage of 05 marks towards the final result.
→ For 5 marks- Diverse methods of assessment as per the need of the class dynamics and curriculum transaction. These may include – short tests, oral test, quiz, concept maps, projects, posters, presentations and enquiry-based scientific investigations etc. and use rubrics for arguing them objectively. This will also have a weightage of 05 marks towards the final result.
b. Practical / Laboratory work should be done throughout the year and the student should maintain record of the same. Practical Assessment should be continuous. There will be weightage of 5 marks towards the final result. All practicals listed in the syllabus must be completed.
c. Portfolio to be prepared by the student- This would include class work and other sample of student work and will carry a weightage of 5 marks towards the final results.
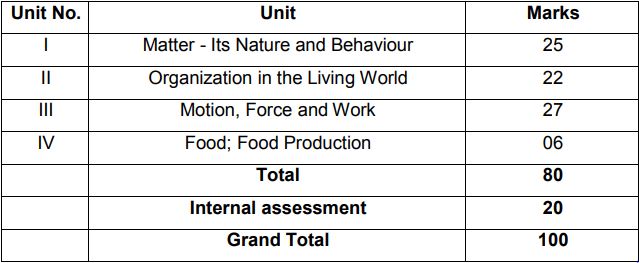
Check Course Structure for Class 9 Science (Annual Examination):
Theme: Materials
Unit I: Matter-Nature and Behaviour
Definition of matter; solid, liquid and gas; characteristics – shape, volume, density; change of statementing (absorption of heat), freezing, evaporation (cooling by evaporation), condensation, sublimation.
Nature of matter: Elements, compounds and mixtures. Heterogeneous and homogenous mixtures, colloids and suspensions. Physical and chemical changes (excluding separating the components of a mixture).
Particle nature and their basic units: Atoms and molecules, Law of Chemical Combination, Chemical formula of common compounds, Atomic and molecular masses.
Structure of atoms: Electrons, protons and neutrons, Valency, Atomic Number and Mass Number, Isotopes and Isobars.
Theme: The World of the Living
Unit II: Organization in the Living World
Cell – Basic Unit of life: Cell as a basic unit of life; prokaryotic and eukaryotic cells, multicellular organisms; cell membrane and cell wall, cell organelles and cell inclusions; chloroplast, mitochondria, vacuoles, endoplasmic reticulum, Golgi apparatus; nucleus, chromosomes – basic structure, number.
Tissues, Organs, Organ System, Organism: Structure and functions of animal and plant tissues (only four types of tissues in animals; Meristematic and Permanent tissues in plants).
Theme: Moving Things, People and Ideas
Unit III: Motion, Force and Work
Motion: Distance and displacement, velocity; uniform and non-uniform motion along a straight line; acceleration, distance-time and velocity-time graphs for uniform motion and uniformly accelerated motion, elementary idea of uniform circular motion.
Force and Newton’s laws: Force and Motion, Newton’s Laws of Motion, Action and Reaction forces, Inertia of a body, Inertia and mass, Momentum, Force and Acceleration.
Gravitation: Gravitation; Universal Law of Gravitation, Force of Gravitation of the earth (gravity), Acceleration due to Gravity; Mass and Weight; Free fall. Floatation: Thrust and Pressure. Archimedes’ Principle; Buoyancy.
Work, Energy and Power: Work done by a Force, Energy, power; Kinetic and Potential energy; Law of conservation of energy (excluding commercial unit of Energy).
Sound: Nature of sound and its propagation in various media, speed of sound, range of hearing in humans; ultrasound; reflection of sound; echo.
Theme: Food
Unit IV: Food Production
Plant and animal breeding and selection for quality improvement and management; Use of fertilizers and manures; Protection from pests and diseases; Organic farming.
Practicals
List of experiments for the practical work to be done in the session 2025-26 can be downloaded from the following link:
Note for the Teachers:
1. The chapter Natural Resources (NCERT Chapter 14) will not be assessed in the year-end examination. However, learners may be assigned to read this chapter and encouraged to prepare a brief write up on any concept of this chapter in their Portfolio. This may be for Internal Assessment and credit may be given for Periodic Assessment/Portfolio.
2. The NCERT text books present information in boxes across the book. These help students to get conceptual clarity. However, the information in these boxes would not be assessed in the year-end examination.
General Instructions for Annual Examination and Internal Assessment:
1. There will be an Annual Examination based on the entire syllabus.
2. The Annual Examination will be of 80 marks and 20 marks weightage shall be for Internal Assessment. 3.For Internal Assessment:
a. There will be Periodic Assessment that would include:
- For 5 marks- Three periodic tests conducted by the school. Average of the best two tests to be taken that will have a weightage of 05 marks towards the final result.
- For 5 marks- Diverse methods of assessment as per the need of the class dynamics and curriculum transaction. These may include – short tests, oral test, quiz, concept maps, projects, posters, presentations and enquiry based scientific investigations etc. and use rubrics for arguing them objectively. This will also have a weightage of 05 marks towards the final result.
b. Practical / Laboratory work should be done throughout the year and the student should maintain record of the same. Practical Assessment should be continuous. There will be weightage of 5 marks towards the final result. All practicals listed in the syllabus must be completed.
c. Portfolio to be prepared by the student- This would include classwork and other sample of student work and will carry a weightage of 5 marks towards the final results.
PRESCRIBED BOOKS:
- Science-Textbook for class IX-NCERT Publication
- Assessment of Practical Skills in Science-Class IX – CBSE Publication
- Laboratory Manual-Science-Class IX, NCERT
- Exemplar Problems Class IX – NCERT Publication